Together, sharing electrons to fill each other's valence shells,Īnd each of the two oxygens form a double bond with carbon. Share two electrons each with another atom so they can have eightĮlectrons in their valence shell and be "full". That it bonds to when it becomes carbon dioxide have six electrons That means it wants other atoms to share fourĮlectrons with it so it can fill its shell. Carbon has four electrons in its valence shell and wants

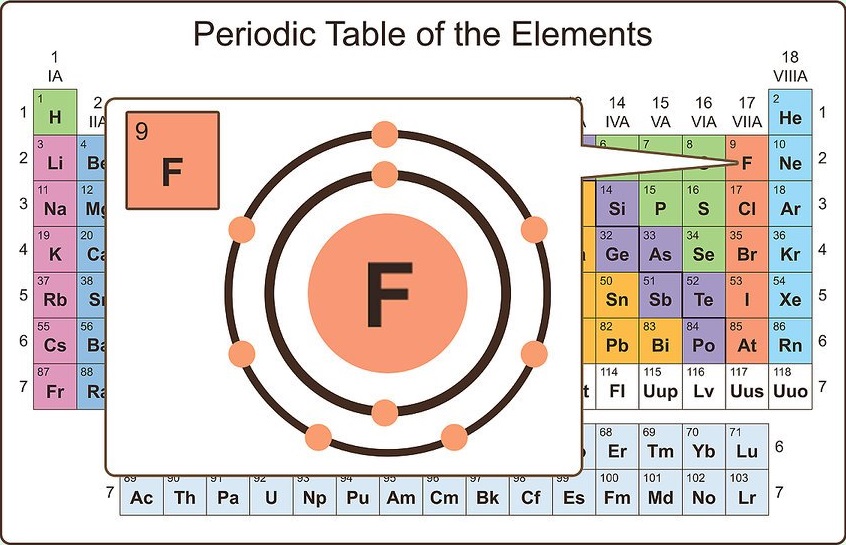
A common example of double bonding is carbonĭioxide. The easiest way possible, using single bonds. Need two electrons to do this, but the other elements need eightĮlectrons in their valence shell. Beginning with beryllium, we see that its nearest preceding noble gas is helium and that the principal quantum number of its valence shell is n 2. He wrote the formula as "O 3B 2".Why would some atoms share more than one pair of electrons?Ītoms are always trying to get a full valence shell (outer shell Solution: The group 2 elements are in the s block of the periodic table, and as group 2 elements, they all have two valence electrons. Stephen was asked to write the formula of the compound formed when boron and oxygen atoms combine. Write the formula for the compound formed between:Ī) calcium and nitrogen (calcium nitride),Ĭ) fluorine and aluminium (aluminium fluoride)į) phosphorus and calcium (calcium phosphide) Write the formula for the compound formed between calcium and carbon called calcium carbide. Lets look at another example by writing the formula of the compound formed between oxygen and aluminium (aluminium oxide) Step 3 Swap the subscripts and divide by the smallest number only if the larger number is divisible by the smaller number. Step 2 Write the valency (without the sign) for each atom as a subscript. Step 1 Write down the two atoms and their valency always putting the atom with the positive valency on the left. The electron configurations of the elements are in Figure 6.9.2. When writing the formula the idea is to create a neutral compound by using as few atoms from each element as possible. 1: The Periodic Table, Showing How the Elements Are Grouped According to the Kind of Subshell ( s, p, d, f) Being Filled with Electrons in the Valence Shell of Each Element. Lithium has a valency of +1 and nitrogen has a valency of -3Ĭombine the atoms of nitrogen and lithium to give the formula of lithium nitride. First we will deal with metal, non-metal interactions. For example take the reaction between sodium and chlorine atoms.Ĭompounds formed between metals and non-metals will be treated differently. Valencies come in really handy when we are looking to write formulae of compounds formed from metal and non-metal atoms. Group 8 elements do not react and so have a valency of 0 Group 7 elements have 7 valence electrons and will tend to take 1 electron and have a valency of -1. Group 6 elements, have 6 valence electrons and will tend to take 2 electrons and have a valency of -2. Group 5 elements, however, have 5 valence electrons and will tend to take 3 electrons and so have a valency of -3. This is the same for group 2 which will give up two electrons and group 3 which will give up 3 electron. The periodic table, shown on the left, can tell us a great deal about the valency of elements.Įlements are placed in groups (columns) in the periodic table according to the number of valence electrons, so naturally the position of the element in the periodic table should give us an idea of its valency.Īll elements in group 1 have 1 valence electron so they have a valency of +1 as they will tend to give up 1 electron. Let's not over complicate this and go straight into the periodic table and how we can tell the valency of an element. The valency of polyatomic ions (such as SO 4 2- ) is the charge on the ion. The valency of an element is determined by the number of outer shell (valence) electrons. Valency refers to the ability of an atom or a group of chemically bonded atoms to form chemical bonds with other atoms or groups of atoms. Ionic compounds are substances made from the combination of charged ( positive and negative) particles. In order to write chemical formulae we need to understand how elements combine with each other to form molecules or ionic compounds.
